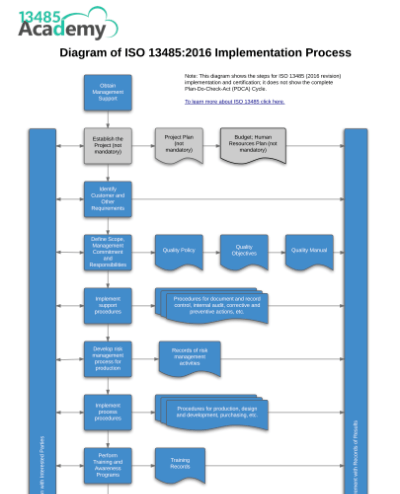 
The framework of this quality management system focuses on the need to handle risk management, in line with ISO 14971. Besides ISO 13485, US FDA 21 CFR Part 820 compliance is also mandatory for companies entering the United States market. 
This covers processes spanning the entire product life-cycle, including design and development, production, warehousing, distribution, installation and servicing activities. The ISO 13485: Quality Management System for Medical Devices is an international standard that was specifically published to define the requirements of a quality management system for organisations within the medical device industry.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
